
SA3800全光谱细胞分析仪
详细介绍
The SA3800 spectral analyzer improves sensitivity and simplifies application design, workflow, and analysis over conventional flow cytometers. This is achieved using spectral analysis technology, automation throughout the system, advanced electronics, and patented optical technologies, unique to Sony systems.
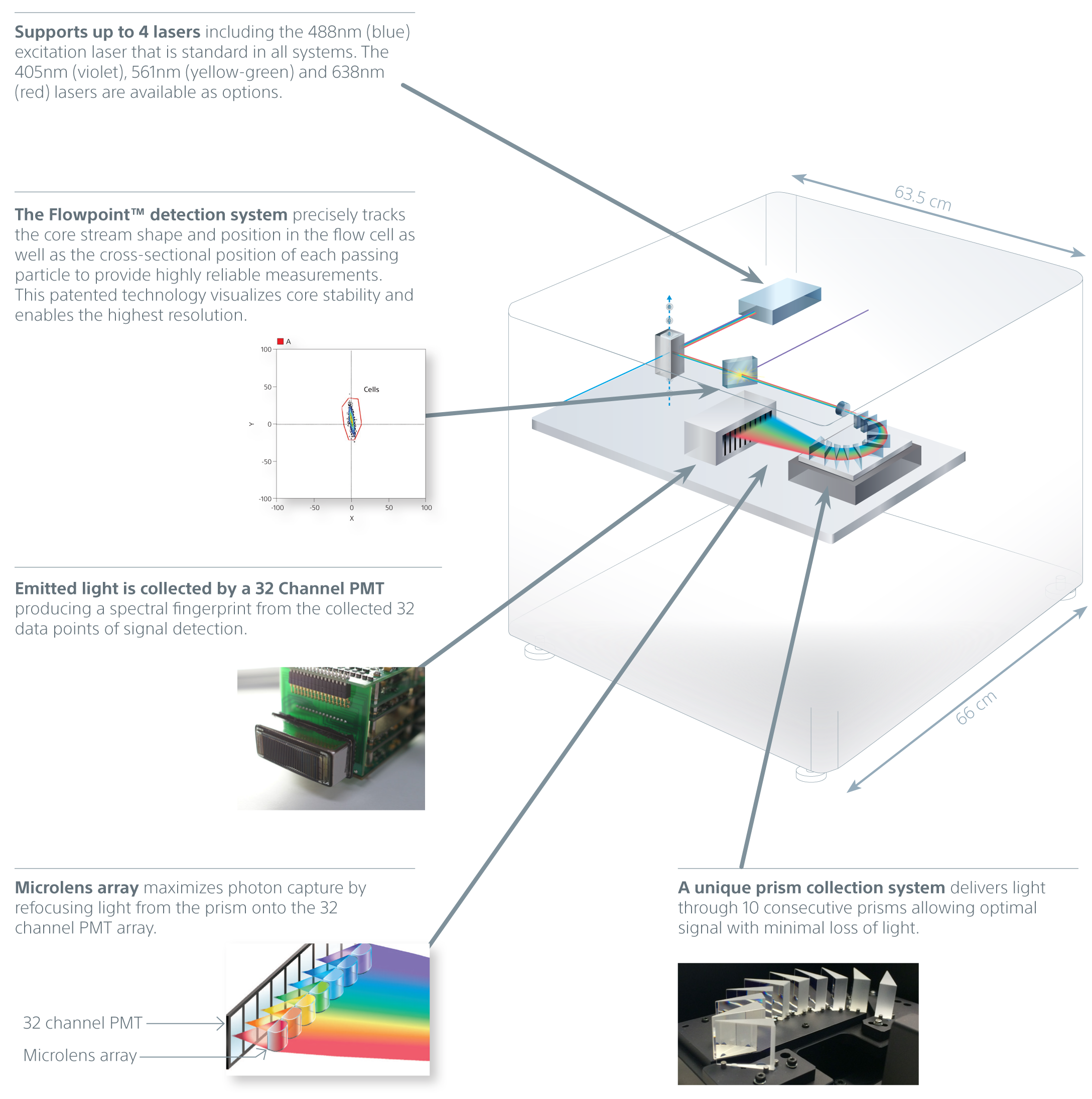
Spectral Analysis Technology
Spectral analysis technology is the foundation of the SA3800 system. Spectral flow cytometry streamlines workflow and yields better data by collecting the entire visual spectrum of light. In conventional systems, overlapping fluorescence is subtracted using color compensation, so less light is collected. Instead, spectral flow cytometers sum the fluorescence together and then use unmixing to mathematically separate the colors. This powerful capability also simplifies workflow, including panel design, and improves visualization of autofluorescence.
A unique prism collection system delivers emitted light to a 32-channel PMT. This produces 34 data points of signal detection for fluorescence and bright autofluorescence to achieve accurate visualizations of fluorescent populations. Researchers can see the complete spectral fingerprint of each fluorochrome from 420 nm to 800 nm.
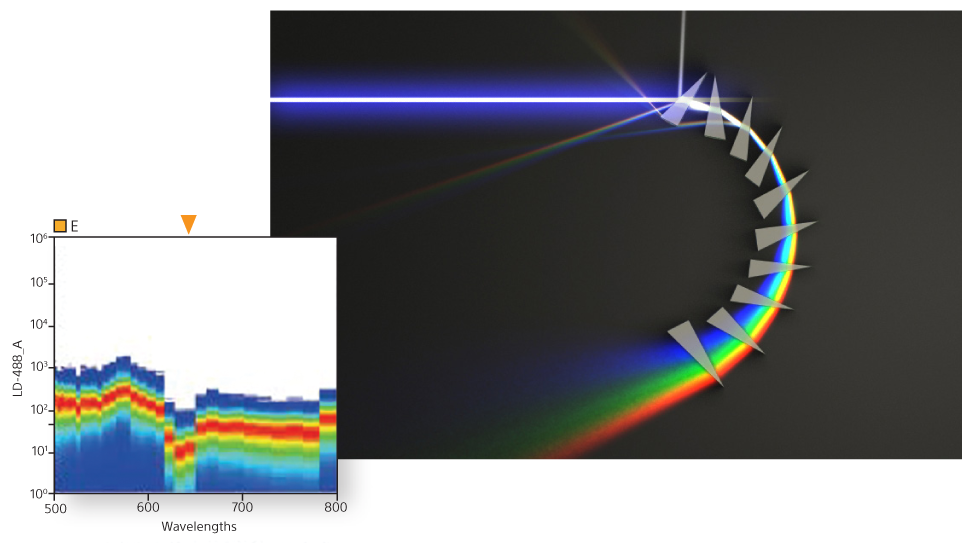
Spectral Unmixing
A powerful capability of spectral technology is unmixing. This technique allows researchers to separate fluorophores into pure signals that measure the quantity of each fluorophore at each pixel to more accurately measure data for analysis.
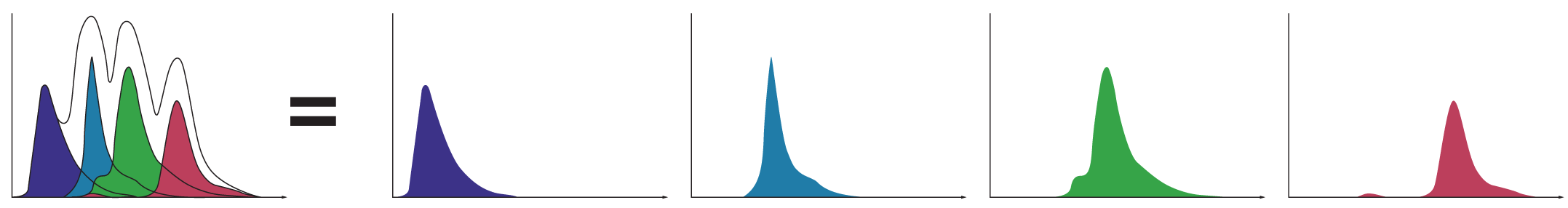
Spectral unmixing separates each spectral fingerprint to better visualize each fluorochrome marker. Unlike conventional filtering in which overlapping signals are lost, spectral unmixing captures the photons emitted from 420 nm to 800 nm. In doing so it enhances dim signal detection for better visualization of rare populations, fluorescent proteins, and fluorochromes excited by multiple lasers.
This also lets researchers separate the spectra of fluorophores masked by autofluorescence by extracting it from the signal and creating a unique autofluorescence parameter. With a clear signal for each color channel unaffected by overlapping signals (spillover) and autofluorescence, spectral analysis yields better, unbiased data for analysis.
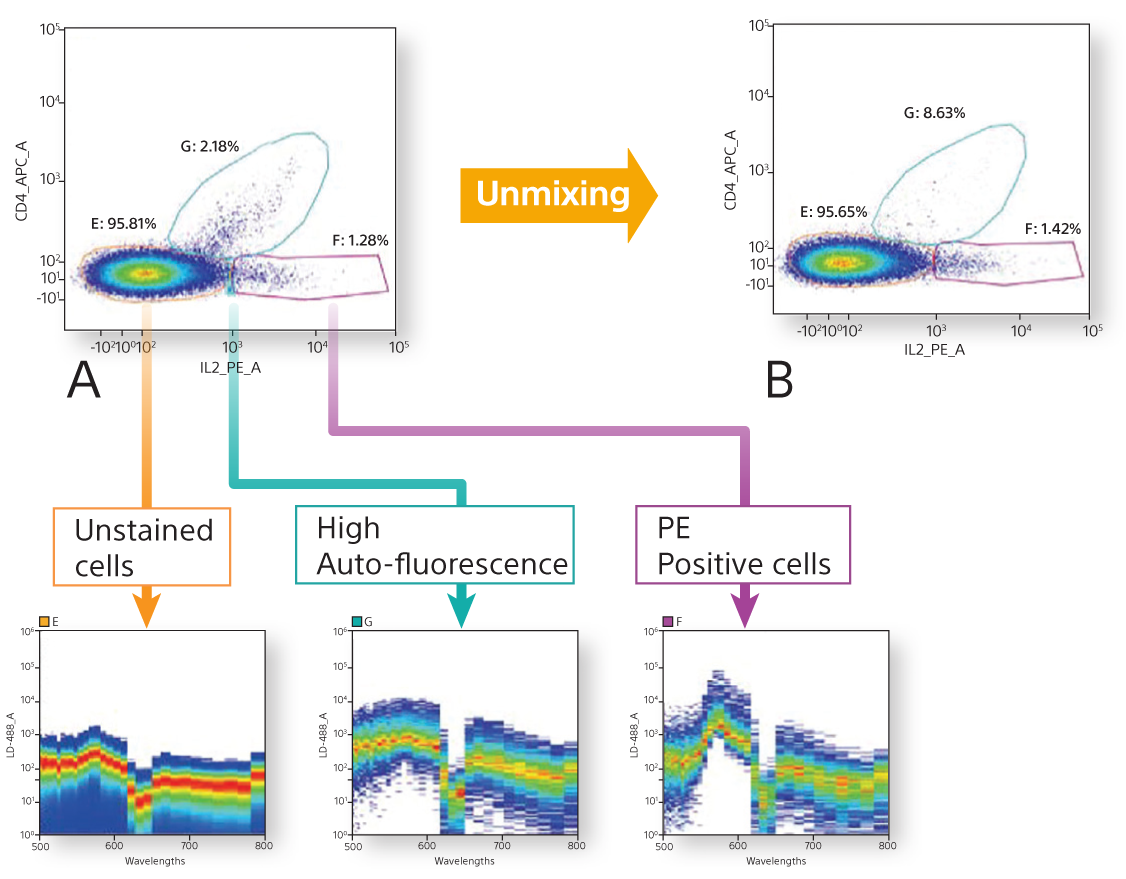
Spectral analysis reduces false positives and delivers more accurate analysis over conventional flow cytometry. Mouse splenocytes were stained with CD4 APC and anti-IL-2 PE. A. In this conventional density plot, it is unclear if the light-blue region is a dim PE, weak double positive, or nonspecific binding. B. Using spectral analysis, the spectral data of each region is compared against the contents of the Spectral Library to unmix the sample. This reveals the light-blue region is high autofluorescence. Representative data collected on the Sony SP6800.
System Automation
Automation is present across the workflow to simplify operation and ensure accurate results. The system supports a wide variety of standard and deep-well plates, and 12 x 75-mm 5-mL tubes in the tube loader.
Novel 3D AutoSampler Technology
The novel 3D AutoSampler uses a fixed probe and moves the plate in horizontal and vertical directions to minimize sample-to-sample cross-contamination and speed up cleaning.
Sensors on the probe enable the system to accurately move the sample to the probe, calculate height of the tube or plate, and automatically recoil the probe if it touches the container or base plate surface. This unique design is aimed at reducing cross-contamination and clogs, and allows for automatic recovery when flow problems are encountered. It also enables faster cleaning between samples.

Plate motion uses a fixed probe and moves the plate (and sample) in horizontal and vertical directions.


The SA3800 supports standard height and 96- and 384-well plates with round, flat, v- and conical shapes in addition to 96-well half deep and bottom plates, 5ml 12x75-mm tubes.
Mixing Automation and Cooling Plate
The 3D AutoSampler mixing function ensures consistent sampling throughout the acquisition of 96- or 384-well sample plates. Software optimizes settings for each plate type. Importantly, the mixing function maintains the integrity and heterogeneity of samples, ensuring that all particles are properly suspended for consistent results.
A cooling plate at the surface of the 3D base further reduces variability and inhibits sample degradation over time.
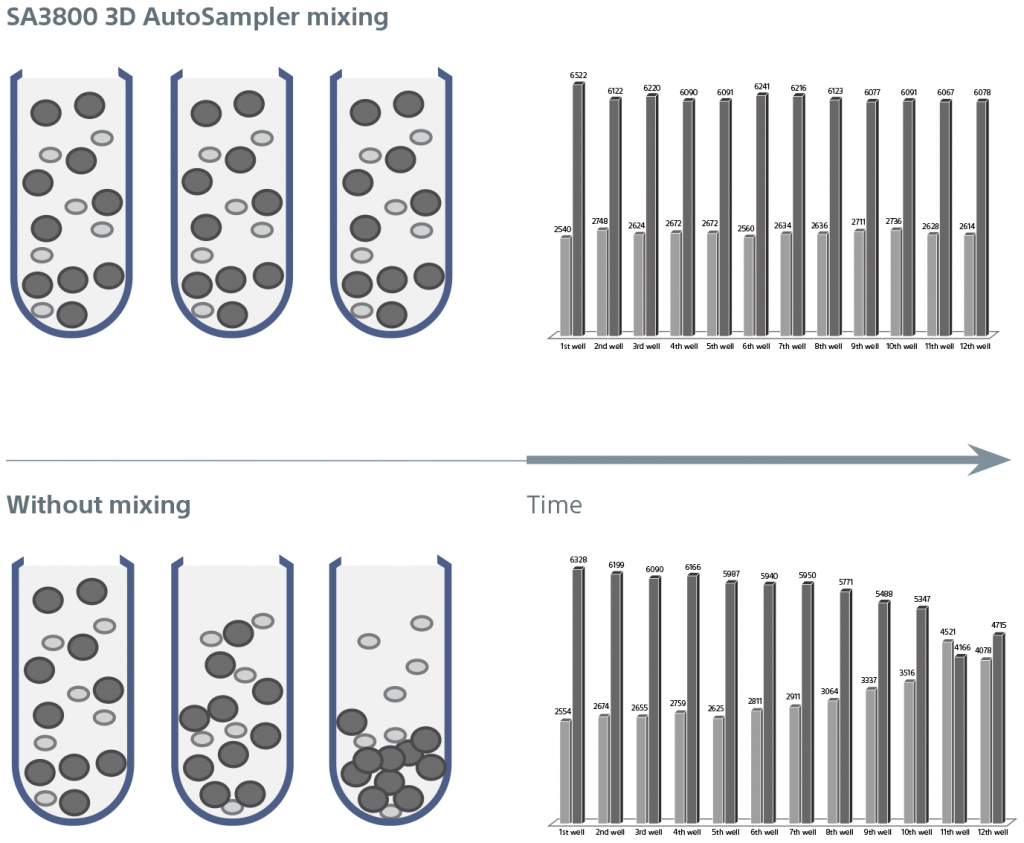
This figure illustrates how cell heterogeneity is maintained using the system’s 3D mixing function. Without mixing, larger cells settle to form heterogeneous samples, as shown in the lower illustration. Graph data describes how the sample integrity is maintained throughout plate acquisition of 3-?m and 10-?m beads from wells 1–96 to deliver more complete, consistent results.
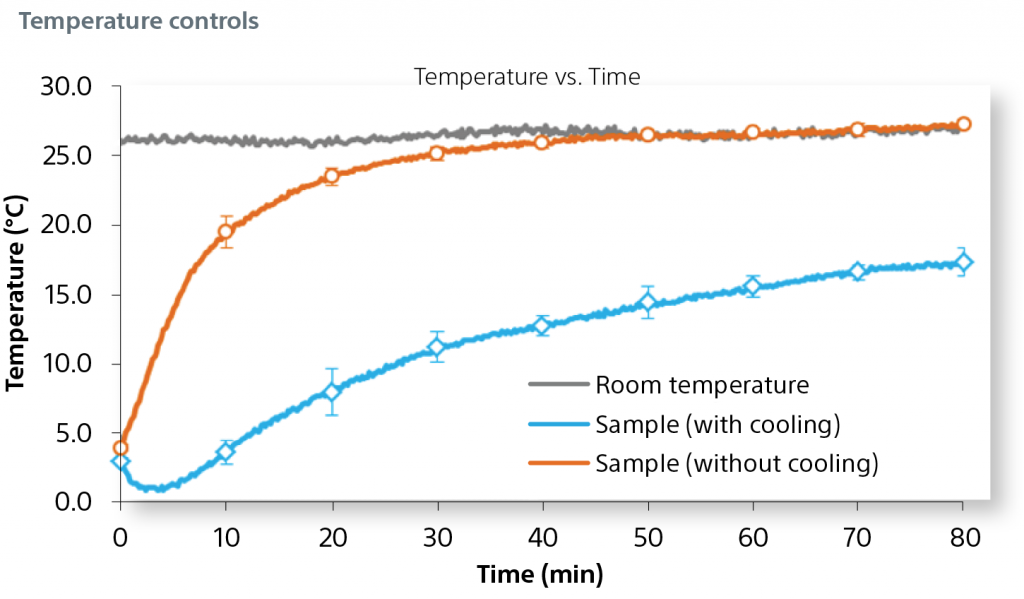
The graph describes the impact of the cooling plate on sample temperature during a 80 minute plate acquisition.
https://www.sonybiotechnology.com/us/instruments/sa3800-spectral-analyzer/system/





 扫描进入手机站
扫描进入手机站